Why Did Oncology Stock Revolution Medicines (RVMD) Gain 41% Monday?
Revolution Medicines (NASDAQ: RVMD) surged over 41 percent after Phase 3 results showed significant survival improvements in pancreatic cancer treatment.
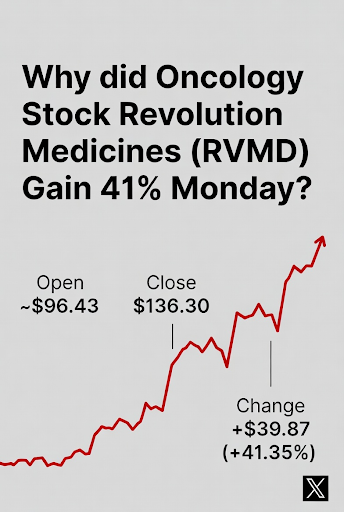
Why Did Oncology Stock Revolution Medicines (RVMD) Gain 41% Monday?
(Investorideas.com Newswire) a go-to platform for big investing ideas, including biotech and medical tech stocks, issues a news and trading alert for Revolution Medicines (Nasdaq: RVMD), a late-stage clinical oncology company developing targeted therapies for patients with RAS-addicted cancers.
Revolution Medicines (Nasdaq: RVMD) was one of the most talked about stocks yesterday as investors realized over 41% in gains as of the close of trading .The stock closed up at 136.30 +39.87 (+41.35%) on volume of over 14 Million shares on news. The average trading volume is 2,440,246.
Yesterday Revolution Medicines announced positive topline results from its global, randomized, controlled Phase 3 RASolute 302 clinical trial evaluating daraxonrasib in patients with metastatic pancreatic ductal adenocarcinoma (PDAC) who had been previously treated. Daraxonrasib taken orally once daily demonstrated statistically significant and clinically meaningful improvements in progression-free survival (PFS) and overall survival (OS) compared with standard of care cytotoxic chemotherapy delivered intravenously. In the overall (intent-to-treat) study population, daraxonrasib demonstrated a median OS of 13.2 months versus 6.7 months for chemotherapy, with a hazard ratio of 0.40 (p < 0.0001). Daraxonrasib was generally well tolerated, with a manageable safety profile and with no new safety signals.
Based on the results from this first interim analysis, all PFS and OS endpoint results are considered final. Revolution Medicines intends to submit these data to global regulatory authorities, including to the U.S. Food and Drug Administration as part of a future New Drug Application under a Commissioner’s National Priority Voucher, and for presentation at the 2026 American Society of Clinical Oncology Annual Meeting.
“For patients with metastatic pancreatic cancer, new treatment options are urgently needed to increase survival time and improve quality of life,” said Brian M. Wolpin, M.D., M.P.H., professor of medicine at Harvard Medical School, director of the Hale Family Center for Pancreatic Cancer Research at Dana-Farber Cancer Institute, and principal investigator for the RASolute 302 trial. “The widely anticipated results of this study indicate that daraxonrasib provides a clear and highly meaningful step forward for patients with pancreatic cancer who have experienced progression on prior treatment, typically chemotherapy. I believe that this new approach is a very important advance for the field that I expect will be practice-changing for physicians and improve the care for patients with previously treated metastatic pancreatic cancer.”
Pancreatic cancer is the most RAS-addicted of all major cancers, with more than 90% of patients harboring tumors driven by mutations in RAS proteins. These mutations span a range of RAS variants that fuel aggressive tumor behavior. Daraxonrasib, a multi-selective inhibitor of RAS(ON) proteins, is the first investigational agent in a novel class of RAS inhibitors designed to address a diverse and broad spectrum of oncogenic RAS drivers.
The RASolute 302 trial enrolled patients with pancreatic tumors harboring a wide range of RAS variants, as well as those without an identified RAS mutation. The primary endpoints of the trial were PFS and OS in patients with tumors harboring RAS G12 mutations. Secondary endpoints assessed PFS and OS in all enrolled patients (the intent-to-treat population), including those with tumors with and without (wild type) an identified RAS mutation.
Revolution Medicines ( Nasdaq: RVMD) is a late-stage clinical oncology company developing novel targeted therapies for patients with RAS-addicted cancers. The company’s R&D pipeline comprises RAS(ON) inhibitors designed to suppress diverse oncogenic variants of RAS proteins. The company’s RAS(ON) inhibitors daraxonrasib (RMC-6236), a RAS(ON) multi-selective inhibitor; elironrasib (RMC-6291), a RAS(ON) G12C-selective inhibitor; zoldonrasib (RMC-9805), a RAS(ON) G12D-selective inhibitor; and RMC-5127, a RAS(ON) G12V-selective inhibitor, are currently in clinical development. Additional development opportunities in the company’s pipeline focus on RAS(ON) mutant-selective inhibitors, including RMC-0708 (Q61H) and RMC-8839 (G13C).
This article is for informational purposes only and is not investment advice. See our full disclaimer.